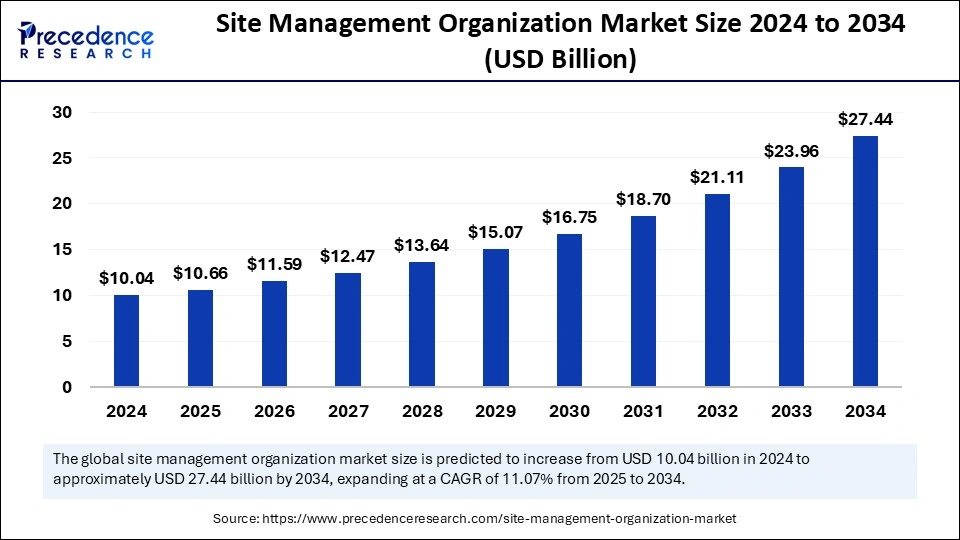
The global site management organization market size is expected to attain around USD 27.44 billion by 2034 increasing from USD 10.04 billion in 2024, with a CAGR of 11.07%.
Site Management Organization Market Key Insights
-
North America captured the largest market share of 42.89% in 2024.
-
Asia Pacific is anticipated to grow at the fastest CAGR during the forecast period.
-
The patient recruitment and retention service segment held the highest share of 23.71% in 2024.
-
The regulatory compliance service segment is expected to expand at the fastest CAGR of 12% over the forecast years.
-
In terms of clinical trial phases, Phase III dominated the market with a 41.64% share in 2024.
-
The Phase II segment is forecasted to grow at a notable CAGR of 10% over the projected period.
-
The oncology segment accounted for the highest share of 33.30% in 2024 within the therapeutic area category.
-
The neurology segment is likely to register a strong CAGR of 10.9% over the study period.
-
The pharmaceutical companies segment held the leading share of 39.26% in 2024 among end-users.
-
The biotechnology companies segment is expected to expand at a remarkable CAGR of 11.4% from 2025 to 2034.
AI Impact on Site Management Organization Market
AI is significantly transforming the Site Management Organization (SMO) market by improving efficiency, reducing costs, and enhancing the overall quality of clinical trial management. AI tools are used to streamline various aspects of site management, from patient recruitment to regulatory compliance and data analysis.
-
Improved Patient Recruitment and Retention
AI-powered systems analyze large volumes of data to identify the best candidates for clinical trials quickly and accurately. This reduces the time spent on manual recruitment processes and increases the likelihood of finding suitable participants. Additionally, AI-driven tools can help personalize engagement strategies, enhancing patient retention rates throughout the trial. -
Optimized Site Selection and Performance
AI assists in selecting the most appropriate clinical trial sites by evaluating historical data, patient demographics, and site performance. This enables SMOs to choose sites with the highest likelihood of success, improving efficiency and reducing trial delays. AI can also predict site performance in real-time, allowing for quick adjustments if necessary. -
Enhanced Operational Efficiency
AI automation streamlines administrative tasks such as data entry, scheduling, and documentation. This not only reduces human errors but also frees up time for site managers and staff to focus on more critical tasks. AI can also assist in regulatory compliance by automatically checking if trial activities meet required standards, thus reducing the risk of non-compliance. -
Real-Time Monitoring and Data Analysis
AI allows for real-time monitoring of clinical trials, ensuring that trials are progressing according to schedule and identifying any potential issues before they become significant problems. AI-driven analytics can analyze patient data, track trial progress, and provide insights into trial outcomes, enabling better decision-making. -
Cost Reduction and Time Efficiency
By automating routine tasks and optimizing site selection, AI significantly reduces operational costs. AI tools can also forecast potential delays and suggest corrective measures, helping to keep trials on track and within budget. This not only improves financial efficiency but also shortens the time required to bring new drugs or treatments to market.
Get a Free Sample Copy of the Report@ https://www.precedenceresearch.com/sample/5762
Market Overview
The Site Management Organization market is growing steadily as pharmaceutical companies seek efficient ways to conduct clinical trials. SMOs help streamline trial operations, ensuring compliance with regulatory standards while improving patient recruitment and retention. As the demand for faster and cost-effective clinical trials increases, the role of SMOs in the research ecosystem becomes more critical.
Market Scope
| Report Coverage | Details |
| Market Size by 2034 | USD 27.44 Billion |
| Market Size in 2025 | USD 10.66 Billion |
| Market Size in 2024 | USD 10.04 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 11.07% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacfic |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Service Type, Phase of Clinical Trials, Therapeutic Area, End-user, and Regions. |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Drivers
The rise in chronic diseases and the demand for innovative therapies are key drivers of the SMO market. Pharmaceutical companies are conducting more trials to bring new treatments to market, increasing the need for well-managed trial sites. Additionally, the growing adoption of digital health solutions, including AI-driven trial monitoring and electronic data capture systems, is enhancing the efficiency of site management operations.
Opportunities
There is a significant opportunity for SMOs to expand their services in emerging markets where clinical trial costs are lower. The adoption of decentralized clinical trials, enabled by telemedicine and remote monitoring technologies, provides another growth avenue. Furthermore, AI and machine learning advancements offer new ways to optimize patient recruitment and site performance, improving trial outcomes.
Challenges
The primary challenges in the SMO market include regulatory hurdles and patient recruitment difficulties. Each country has its own set of regulations for clinical trials, requiring SMOs to navigate complex approval processes. Additionally, retaining participants throughout long clinical trials can be difficult, impacting trial timelines and success rates. High operational costs also remain a concern, particularly for smaller SMOs.
Regional Insights
North America leads the market due to its strong clinical trial infrastructure and regulatory framework. Europe is another major player, benefiting from supportive regulations and advanced research facilities. The Asia-Pacific region is growing rapidly, driven by increasing clinical trial activities, a diverse patient population, and cost advantages. Latin America and the Middle East are also emerging as attractive locations for trial expansion due to their growing healthcare infrastructure.
Recent Development
- In March 2025, Neutra announced the launch of its Site Management Organization (SMO), Neutra Life Sciences. This new organization is set to improve the efficiency, quality, and scalability of clinical trials, ensuring seamless execution and improved outcomes for sponsors and research institutions worldwide.
Site Management Organization Market Companies

- Novotech
- ERG Holding
- Apex Medical Research
- CMIC Group
- Tigermed
- FORMAT Medical Research
- EPSI
- AusTrials
- Beijing Aisimo Medical Science and Technology Co.
- Ltd
- MEDEX
- Ethic Co.
- ACTG-CRO
- CIDAL
- MPR Development Group
Segments Covered in the Report
By Service Type
- Site selection
- Patient recruitment & retention
- Site training & support
- Data management
- Regulatory compliance
- Monitoring services
- Project management
By Phase of Clinical Trials
- Phase I
- Phase II
- Phase III
- Phase IV
By Therapeutic Area
- Oncology
- Cardiovascular
- Neurology
- Infectious diseases
- Metabolic disorders
- Others
By End-user
- Pharmaceutical companies
- Biotechnology companies
- Academic institutions
- Government & non-profit organizations
By Geography
- North America
- Asia Pacific
- Europe
- Latin America
- Middle East & Africa
Also Read: Raman Spectroscopy Market
Ready for more? Dive into the full experience on our website@ https://www.precedenceresearch.com/