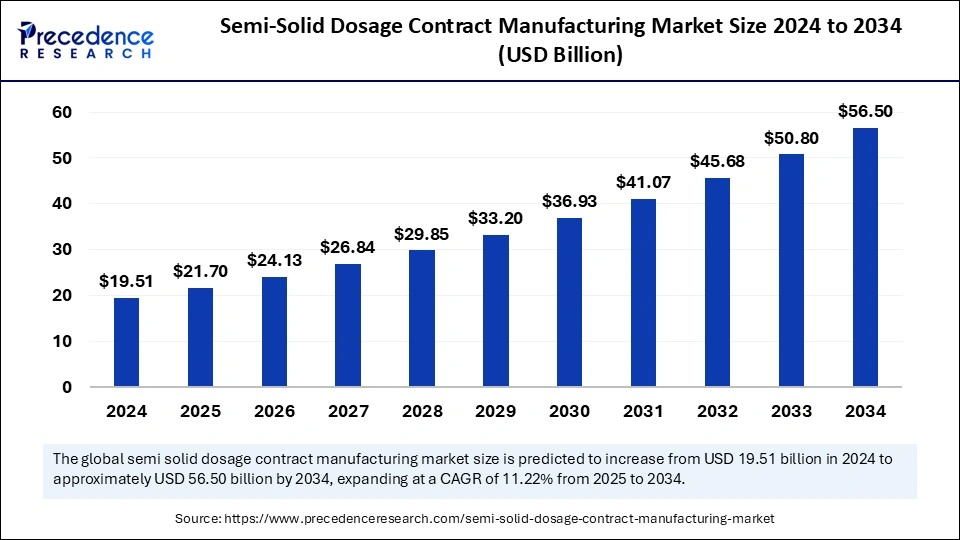
The global semi-solid dosage contract manufacturing market size was valued at USD 19.51 billion in 2024 and is projected to attain around USD 56.50 billion by 2034, growing at a CAGR of 11.22%.
The semi-solid dosage contract manufacturing market is growing as pharmaceutical companies look for flexible and cost-effective production solutions for products like creams, ointments, and lotions. Outsourcing manufacturing operations allows pharmaceutical firms to leverage the expertise of specialized CMOs, ensuring product consistency and regulatory compliance. The increasing focus on dermatological and transdermal drug formulations is further boosting market demand.
Semi-Solid Dosage Contract Manufacturing Market Key Highlights
-
Asia Pacific secured the highest market share, exceeding 33% in 2024.
-
North America is expected to grow at a CAGR of 10.67% in the forecast period.
-
The topical segment led the market, holding 50% of the total share in 2024.
-
The transdermal segment is set to grow steadily over the projected years.
-
Creams were the leading product category in 2024.
-
Large-sized companies commanded a dominant market position in 2024.
-
The medium and small-sized companies segment is anticipated to expand significantly.
-
Pharmaceutical companies emerged as the top end-user segment in 2024.
-
Cosmeceutical companies are predicted to experience steady growth during the forecast period.
Role of AI in Semi-Solid Dosage Contract Manufacturing Market
AI plays a pivotal role in the Semi-Solid Dosage Contract Manufacturing Market by enhancing various aspects of the manufacturing process, improving efficiency, and ensuring higher quality in semi-solid formulations. AI technologies such as machine learning, data analytics, and automation systems are being integrated into production lines to optimize formulation development, reduce errors, and minimize production downtime.
In the development phase, AI helps in analyzing large volumes of data to identify optimal ingredient combinations, improve formulation stability, and predict the behavior of different semi-solid dosage forms. It enables faster product development by automating repetitive tasks and optimizing lab processes. AI-powered predictive models can anticipate issues related to formulation consistency and stability, reducing the need for extensive trial-and-error testing.
AI also plays a crucial role in enhancing quality control. Through machine vision systems and automated inspection tools, AI can identify defects in packaging, check for contamination, and ensure that dosage levels are consistent across batches. Additionally, AI systems can analyze real-time production data to provide valuable insights, helping manufacturers to adjust processes quickly and ensure compliance with Good Manufacturing Practices (GMP) standards.
Furthermore, AI-driven automation in production processes helps reduce human intervention, increasing production speed and reducing costs. It also supports predictive maintenance of manufacturing equipment, reducing the likelihood of breakdowns and improving operational uptime. As the demand for high-quality, consistent semi-solid dosage forms rises, AI’s role in streamlining operations and improving product quality is becoming increasingly indispensable.
What is Semi-Solid Dosage Contract Manufacturing?
Semi-solid dosage contract manufacturing refers to the outsourcing of production for pharmaceutical and cosmeceutical products in semi-solid forms, such as creams, gels, ointments, pastes, and lotions. These dosage forms are commonly used for dermatological, transdermal, and mucosal drug delivery applications. Pharmaceutical companies often partner with contract manufacturers to leverage their expertise, reduce costs, and ensure compliance with regulatory standards.
Market Growth and Demand
The demand for semi-solid dosage forms is growing due to the increasing prevalence of skin-related diseases, the rising need for transdermal drug delivery, and advancements in formulation technologies. Contract manufacturers play a crucial role in helping pharmaceutical companies meet market demands while focusing on research and development. The market is expanding as companies seek efficient and cost-effective solutions for product development, testing, and large-scale manufacturing.
Key Services Offered
Contract manufacturers provide a wide range of services, including:
-
Formulation Development: Optimizing the composition of semi-solid drugs for stability and efficacy.
-
Analytical Testing: Ensuring product quality, safety, and compliance with regulatory standards.
-
Manufacturing and Packaging: Large-scale production with high precision and compliance with Good Manufacturing Practices (GMP).
-
Regulatory Support: Assisting pharmaceutical companies in meeting FDA, EMA, and other global regulatory requirements.
Technological Advancements
AI, automation, and advanced formulation technologies are revolutionizing semi-solid dosage contract manufacturing. AI-driven predictive analytics help optimize formulations, while automation enhances precision and reduces human errors. Innovations such as nanoemulsions, lipid-based drug delivery, and controlled-release formulations are improving the efficiency and effectiveness of semi-solid drugs.
Regulatory Considerations
The industry is highly regulated, with strict guidelines from agencies like the U.S. FDA, the European Medicines Agency (EMA), and other regulatory bodies worldwide. Compliance with GMP, stability testing, and quality assurance protocols is essential for contract manufacturers to ensure product safety and efficacy.
Market Scope
| Report Coverage | Details |
| Market Size by 2034 | USD 56.50 Billion |
| Market Size in 2025 | USD 21.70 Billion |
| Market Size in 2024 | USD 19.51 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 11.22% |
| Dominated Region | Asia Pacific |
| Fastest Growing Market | North America |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Type, Product, Company Size, End-use, and Regions |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Market Dynamics
Market Drivers
The market is being driven by the rising incidence of chronic skin conditions, increasing demand for over-the-counter (OTC) topical treatments, and advancements in formulation technologies. Additionally, contract manufacturing helps pharmaceutical companies reduce capital expenditures on production facilities, enabling them to invest more in research and development. The growing geriatric population, which requires long-term dermatological treatments, is also fueling market expansion.
Market Opportunities
Opportunities in the semi-solid dosage contract manufacturing market include the expansion of pharmaceutical companies into emerging markets, the development of nano-formulations for enhanced drug delivery, and the increasing use of AI-driven quality control systems. Additionally, the growing demand for prescription and OTC semi-solid drugs in the cosmetics and skincare industry is creating new business prospects for CMOs.
Market Challenges
Despite the market’s growth, regulatory hurdles remain a major challenge, as different countries impose varying compliance standards on drug manufacturing. The risk of contamination and stability issues in semi-solid formulations also poses challenges for manufacturers. Furthermore, price competition among CMOs can pressure profit margins, making it necessary for companies to differentiate through innovation and efficiency.