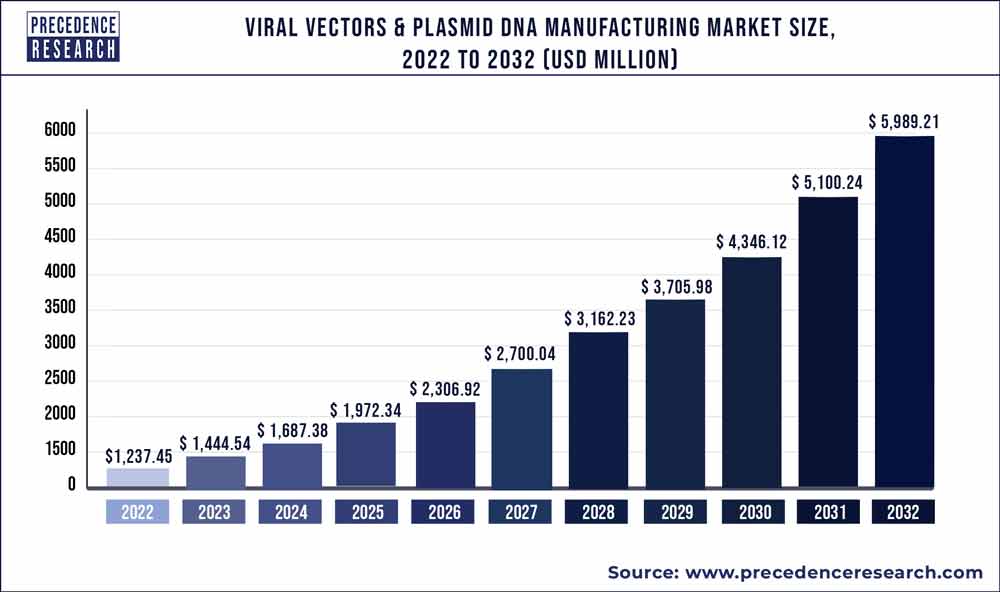
The global viral vectors & plasmid DNA manufacturing market was valued at USD 1,237.45 million in 2022 and is expected to reach USD 5,989.21 million by 2032, noteworthy a CAGR of 16.8% from 2023 to 2032.
Key Takeaways
- North America is expected to dominate in the advanced IC substrates market during the forecast period.
- By Vector Type, the AAV segment shows a leading growth in the viral vectors & plasmid DNA manufacturing market.
- By Workflow, the downstream processing segment shows a dominant position in the viral vectors & plasmid DNA manufacturing market.
- By Disease, the cancer segment dominated the market and is expected to maintain its dominance during the forecast period.
Drivers
- Rising prevalence of cancer and rare genetic diseases
- Growing research & development of gene therapies in cancer treatment
Market Restraints
- Challenges in executing gene therapies
- High cost associated with gene therapies
Opportunities
- Investment and collaboration in cell and gene therapy sector
- Increase in demand for synthetic genes
Gene therapy is an experimental treatment that comprises incorporating genetic material inside a person’s cells to stopor fight a particular disease. Researchers are studying gene therapy for several diseases including hemophilia, cancer, Parkinson’s disease, severe combined immuno-deficiencies, HIV, via numerous dissimilar methods. A gene can be carried to a cell with the help of a carrier known as a vector. Viruses are the most generalkinds of vectors used in gene therapy. Since last few years, several non-viral and viral vectors have been standardized and enhanced.
Get the Sample Pages of Report@ https://www.precedenceresearch.com/sample/1012
At present, the much wide spread viral vectors utilized for gene therapies are those based on lentivirus, retrovirus, adenovirus, and AAV vectors. These correspondingly form 8%, 16%, 20%, and 8% of the clinical trialsof active gene therapy. Likewise, plasmid DNA has appeared as the most frequently preferred vectors amid non-viral gene delivery tools. Further, it finds application in production and development of DNA vaccines and viral vectors. There is growing demand for practical manufacturing solutions for viral vectors that can be freely scaled and boosted. This is because gene and cell therapies possess latent to quickly advance via clinical trials to commercialization.With the help of ongoing efforts, several organizations are respectivelyexecutingand developing state-of-the-art solutions intended to scale-up for viral vector manufacturing and speed up process development.
Global Market Revenue (USD Million) and Growth Rate Comparison by Workflow (2016-2027)
| Workflow | 2016 | 2019 | 2027 | CAGR % (2020-2027) | |
| Upstream Processing | 94.60 | 165.45 | 770.72 | 21.27 | % |
| Downstream Processing | 145.60 | 261.13 | 1,301.65 | 22.30 | % |
| Total | 240.20 | 426.58 | 2,072.36 | 21.91 | % |
Growth Factors
Viral vectors have become ideal choice for gene transferdue to their efficient gene delivery, high transfection efficiency and stable gene expression. Further, upsurge in registration of clinical trials on viral vector-mediated gene therapy is stimulating inclination for viral vectors in gene transfer. Growing pervasiveness of target disorders and diseases, the accessibility of funding for gene therapy development, current research into viral vector-based cell and gene therapies and efficacy of viral vectors in gene therapy delivery are together supporting the market growth. Unexploitedlatent in emergent markets is projected to provideworthwhile growth opportunities for participants in this market. Furthermore, groundbreaking development in the vaccinology is fuelling the demand of these vectors to greater extent. Great amount of clinical and preclinical studies assessing the prospective of vectors in these cutting-edge therapies haveexhibitedfavorable results. As a result of this,several investors are attracted towards this area which is making plasmid and viral vector manufacturing market avigoroussector of investment. Further, it is inspiringfortunate funding activities from both the public and private sectors.
Report Highlights
- By Vector Type, in 2019, AAV dominated the market with around 22.83% share in terms of revenue of the total market. Lentivirus is projected to grow at highest CAGR of 23.27% during forecast period and anticipated to attain a market share of 17.41% in 2027.
- The global market for the Plasmid DNA was valued at USD 83.5 million in 2019 and is expected to reach USD 407.4 million by 2027.
- By Disease Type, in 2019, cancer dominated the market with around 55.32% share in terms of revenue of the total market. This growth is credited to the rising cancer incidence across the globe. Cancer is also projected to grow at highest CAGR of 22.32% during forecast period.
- By Application Type, in 2019, gene therapy dominated the market with around 37.23% share in terms of revenue of the total market. The global market for the Antisense &RNAi was valued at USD 111.5 million in 2019 and is expected to reach USD 543.4 million by 2027.
- By End-User, in 2019, Biopharmaceutical & Pharmaceutical Companies dominated the market with around 67.45% share in terms of revenue of the total market. Biopharmaceutical & Pharmaceutical Companies is projected to grow at highest CAGR of 22.27% during forecast period.
- Europe Viral Vectors & Plasmid DNA Manufacturing market was estimated at USD 92.7 million in 2019 and is projected to reach value USD 435.2 million by 2027.
- Asia Pacific Viral Vectors & Plasmid DNA Manufacturing market was estimated at USD 56.2 million in 2019 and is projected to reach value USD 294.5 million by 2027.
Key Players & Strategies
Several established organizations have been involved in the production of vectors since the inception of this domain. However, the growing demand for these programs has spurred the establishment of many start-ups as well. Examples include (indicative list, in alphabetical order) Batavia Biosciences, Brammer Bio, GenIBET Biopharmaceuticals, Immune Technology, Lentigen Technology, Luminous Biosciences, Oxford Genetics, SignaGen Laboratories, Vectalys and Virovek. It is also worth highlighting that over 50 academic institutes / non-profit organizations are currently involved in the production of vectors for use in gene therapies.
The demand for clinical grade and research vectors is much than commercial grade vectors and almost many gene therapy entrants are in development stage. Nevertheless, some companieshave are rigorouslymanufacturing commercial scale capacity for vector production. Some of these players includeBioReliance, Aldevron, Eurogentec, Cobra Biologics, Lonza, MassBiologics, FUJIFILM Diosynth Biotechnologies and WuXiAppTec amongst others.
Read Also: Smart Healthcare Market Size to Hit US$ 1,013.14 Bn by 2032
Key Market Players:
- Novasep
- Aldevron
- MerckWaismanBiomanufacturing
- Creative Biogene
- The Cell and Gene Therapy Catapult
- Cobra Biologics
- uniQure N.V.
- Addgene
- FUJIFILM Holdings Corporation
- Oxford Biomedicaplc
- Takara Bio Inc.
Market Segmentation:
By Vector Type
- Adenovirus
- Plasmid DNA
- Lentivirus
- Retrovirus
- AAV
- Others
By Application
- Gene Therapy
- Antisense &RNAi
- Cell Therapy
- Vaccinology
By Workflow
- Upstream Processing
- Vector Recovery/Harvesting
- Vector Amplification & Expansion
- Downstream Processing
- Fill-finish
- Purification
By End-User
- Biopharmaceutical and Pharmaceutical Companies
- Research Institutes
By Disease
- Genetic Disorders
- Cancer
- Infectious Diseases
- Others
By Geography
- North America
- U.S.
- Canada
- Europe
- U.K.
- Germany
- France
- Asia Pacific
- China
- India
- Japan
- South Korea
- Rest of the World
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com
Blog: https://www.expresswebwire.com/
Blog: https://www.uswebwire.com/
- Esoteric Testing Market Size to Hit USD 75.54 Bn by 2032 - July 17, 2024
- mRNA Therapeutics Market Size to Surpass USD 39.99 Bn By 2033 - July 17, 2024
- Acne Treatment Market Size to Hit USD 15.86 Bn by 2033 - July 17, 2024

